Activation of Hypoxia Signaling Pathway Enhances Bone Health and Metabolism in Obesity
Researchers show that pharmacological activation of hypoxia signaling improves metabolism, limits fat accumulation, and restores bone health in obese mice
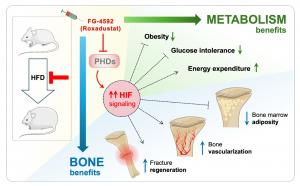
CHENGDU, SICHUAN, CHINA, March 17, 2026 /EINPresswire.com/ -- Obesity harms not only metabolism but also bone strength and repair. This may be attributed to impaired glucose homeostasis and altered bone properties, features that are both strongly affected by hypoxia-inducible factor (HIF) signaling. Now, researchers have evaluated the effect of HIF activation using Roxadustat on bone health and metabolism in obese mice. They observed that Roxadustat prevented weight gain, improved glucose tolerance, and enhanced bone healing, highlighting a therapeutic strategy to tackle both metabolic and skeletal deterioration in obesity.Obesity is widely known for increasing the risk of diabetes and cardiovascular disease, but its damaging effects on the skeleton are often overlooked. Excess body fat can disrupt bone metabolism, weaken bone quality, and impair fracture repair. In individuals with metabolic disorders, bone marrow can accumulate fat cells that interfere with the activity of bone-forming cells and damage the vascular networks that support skeletal tissue. These changes increase fracture risk and reduce the body’s ability to regenerate bone after injury. Despite these clinical challenges, the biological mechanisms connecting metabolic disease and bone health have remained poorly understood.
To explore this connection, a team of researchers led by Professor Christa Maes from the Laboratory of Skeletal Cell Biology and Physiology, KU Leuven, Belgium, investigated the role of hypoxia-inducible factor (HIF) signaling, a molecular pathway that enables cells to adapt to low-oxygen environments while regulating metabolism, blood vessel formation, and tissue repair. Using a mouse model of high-fat diet (HFD)-induced obesity, the team administered the HIF-prolyl-hydroxylase-domain enzyme (PHD) inhibitor FG-4592 (Roxadustat), a drug already approved for treating certain forms of anemia. Mice were fed HFD to mimic metabolic stress, allowing the researchers to examine whether activating hypoxia signaling could improve both metabolic health and skeletal integrity under obesogenic conditions. Their findings were published in Volume 14 of the journal Bone Research on February 11, 2026.
“We hypothesize that activation of the HIF pathway might be an effective therapeutic strategy to enhance both metabolism and skeletal integrity during metabolic stress,” says Prof. Maes.
The experiments revealed striking metabolic benefits in the treated animals. Activation of the hypoxia signaling pathway significantly reduced body-weight gain and limited the accumulation of peripheral fat despite continued exposure to HFD. The treated mice also showed improved glucose tolerance, indicating better control of blood sugar levels. Researchers found that these metabolic improvements were associated with increased energy expenditure, suggesting that the intervention helped the body burn more energy rather than storing it as fat.
Equally important were the protective effects on bone tissue. Obesity often leads to excessive accumulation of adipocytes in bone marrow, which disrupts the balance between bone formation and fat storage. In the treated mice, activation of HIF signaling prevented this abnormal buildup of marrow fat while preserving the vascular network within bone. Maintaining this blood vessel system is essential because it supplies oxygen, nutrients, and molecular signals that support bone maintenance and regeneration.
The researchers also examined fracture healing under metabolic stress. Normally, obesity and impaired glucose metabolism slow down bone regeneration and lead to weaker or even failing repair after injury. However, mice treated with Roxadustat displayed improved fracture healing compared with untreated obese animals that showed compromised bone repair, demonstrating that activation of hypoxia signaling can restore the bone’s regenerative potential.
“Our findings show that activation of hypoxia signaling using PHD inhibitors has dual beneficial effects, simultaneously improving metabolism and bone health,” says Prof. Maes.
Beyond the experimental findings, the work highlights potential ripple effects across multiple research fields. The hypoxia signaling pathway regulates energy metabolism, vascular biology, and tissue regeneration, making it relevant to studies of diabetes, aging, and regenerative medicine.
In the short term, the findings suggest that therapies activating hypoxia signaling could help reduce bone complications in people with obesity or prediabetes. Over the longer term, such approaches may contribute to treatments that simultaneously manage metabolic disease and strengthen skeletal resilience. If similar effects are confirmed in humans, future therapies could reduce fracture risk, improve recovery after bone injuries, and address metabolic dysfunction in a single integrated strategy.
***
Reference
Title of original paper: Pharmacological HIF activation protects against diet-induced obesity, glucose intolerance, and skeletal dysfunction by exerting dual beneficial effects on energy metabolism and bone
Journal: Bone Research
DOI: 10.1038/s41413-025-00503-3
About KU Leuven
KU Leuven is one of Europe’s leading research universities, renowned for excellence in science, medicine, and engineering. Founded in 1425 and located in Leuven, Belgium, the university combines centuries of academic tradition with cutting-edge innovation and global collaboration. KU Leuven is internationally recognized for its strong interdisciplinary research environment and close partnerships with hospitals, research institutes, and industry partners. Its biomedical sciences programs play a major role in advancing translational research aimed at improving human health, disease prevention, and treatment worldwide. The university consistently ranks among the top institutions globally for scientific impact and high-quality research output.
Website: https://www.kuleuven.be/english/kuleuven/
About Professor Christa Maes from KU Leuven
Dr. Christa Maes is a Professor at KU Leuven and group leader of the Laboratory of Skeletal Cell Biology and Physiology (SCEBP lab), which she founded with the support from a European Research Council (ERC) Grant after her postdoctoral research with Henry Kronenberg at Harvard Medical School. Her research focuses on the biology of skeletal stem cells and osteoblasts and how they function to regulate bone homeostasis, bone regeneration, and metabolic control of bone health and disease, with a special interest in how the bone cells crosstalk with the vasculature and other cells and organ systems within and beyond the bone environment. Her team’s contributions include new mechanisms by which bone cells regulate hematopoiesis and interplay with systemic energy metabolism and glucose homeostasis. She has been felicitated with several awards, including the Iain T Boyle Career Award of the European Calcified Tissue Society (ECTS), and she is an active contributer to the bone research communities’ training programs, conference organizations, and journal and book publications and editorial work.
Funding information
This research was supported by grants from the Research Foundation Flanders (FWO) to Christa Maes (#G0B5219N; #G078923N). During the study, Roger Valle-Tenney received an FWO postdoctoral fellowship (#1291522N), and Karen De Samblancx was supported by a FWO PhD fellowship (##1S03417N). Nicolas Peredo was an Early-Stage Researcher in the EU Horizon 2020 research and innovation program under Marie Sklodowska-Curie (MSCA-ITN #721432). µCT equipment at the KU Leuven Department of Development and Regeneration was financed by the Hercules Foundation project AKUL/13/47 (#G0F2414N).
Yini Bao
International Journal of Oral Science
+862885546461 ext.
br@scu.edu.cn
Visit us on social media:
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.